THE NEED FOR REBLOZYL: REBLOZYL MOA
REBLOZYL works to restore balance in erythropoiesis1-4
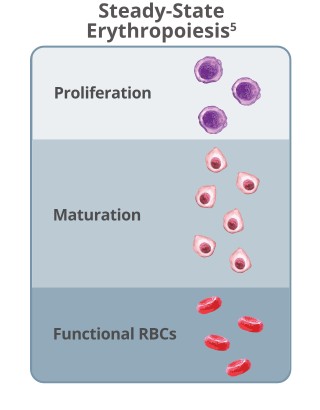

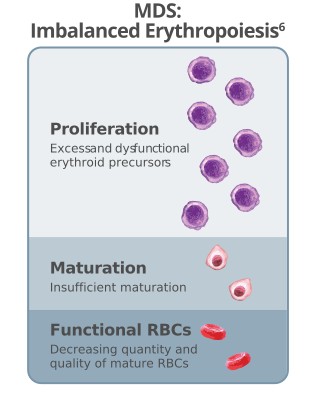



Imbalanced erythropoiesis leads to a lack of mature RBCs and triggers overproduction of erythropoietin.8


REBLOZYL restores erythropoiesis by increasing the number and improving the quality of mature RBCs as observed in preclinical studies3,4
In preclinical models, REBLOZYL improved3,4:
- Hgb levels
- RBC morphology
- Other hematologic parameters† associated with ineffective erythropoiesis
*REBLOZYL binds several TGF-β superfamily ligands, thereby diminishing Smad 2/3 signaling and increasing the number of mature RBCs.2,3
†Other hematologic parameters include reducing oxidative stress in erythrocytes, reducing accumulation of α-globin aggregates in erythrocyte membranes, and improving RBC lifespan.3,4
ESA=erythropoiesis-stimulating agent; Hgb=hemoglobin; MOA=mechanism of action; RBC=red blood cell; TGF-beta=transforming growth factor beta.
1. REBLOZYL [Prescribing Information]. Summit, NJ: Celgene Corporation; 2024. 2. Suragani RNVS, Cadena SM, Cawley SM, et al. Transforming growth factor-β superfamily ligand trap ACE-536 corrects anemia by promoting late-stage erythropoiesis. Nat Med. 2014;20(4):408-414. 3. Attie KM, Allison MJ, McClure T, et al. A phase 1 study of ACE-536, a regulator of erythroid differentiation, in healthy volunteers. Am J Hematol. 2014;89(7):766-770. 4. Suragani RNVS, Cawley SM, Li R, et al. Modified activin receptor IIB ligand trap mitigates ineffective erythropoiesis and disease complications in murine β-thalassemia. Blood. 2014;123(25):3864-3872. 5. Oikonomidou PR, Rivella S. What can we learn from ineffective erythropoiesis in thalassemia? Blood Rev. 2018;32(2):130-143. doi:10.1016/j.blre.2017.10.001. 6. Gupta R, Musallam KM, Taher AT, Rivella S. Ineffective erythropoiesis: anemia and iron overload. Hematol Oncol Clin North Am. 2018;32(2):213-221.doi:10.1016/j.hoc.2017.11.009. 7. Hayati S, Platzbecker U, Aluri S, et al. Luspatercept improves hematopoiesis in lower-risk myelodysplastic syndromes: comparative biomarker analysis of ring sideroblast-positive and -negative subgroups from the phase 3 COMMANDS study. Presented at: European Hematology Association (EHA) Hybrid Congress. June 13-16, 2024, Madrid, Spain. 8. Liang R, Ghaffari S. Advances in understanding the mechanisms of erythropoiesis in homeostasis and disease. Br J Haematol. 2016;174(5):661-673. doi:10.1111/bjh.14194

